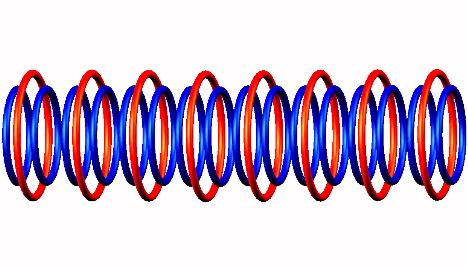
Nuclear structure of (stable). Move mouse over image for Oxygen 17 (stable) nuclear structure
Blue toruses = 16 protons
Red toruses = 8 nuclear electrons
Atomic number of O16 = protons (16) - nuclear electrons (8) = 8 with structure 8D in a line.
Click on image for Oxygen 16 larger image
Photon model of light
J.Lucas, O16 nucleus model
Oxygen 16 isotope stable nucleus natural abundance 99.762 % principal product of stellar evolution



The Oxygen 16 isotope has a highly symmetric stable nucleus, saturated with binding electrons, natural abundance 99.762 %. A principal product of stellar evolution.



