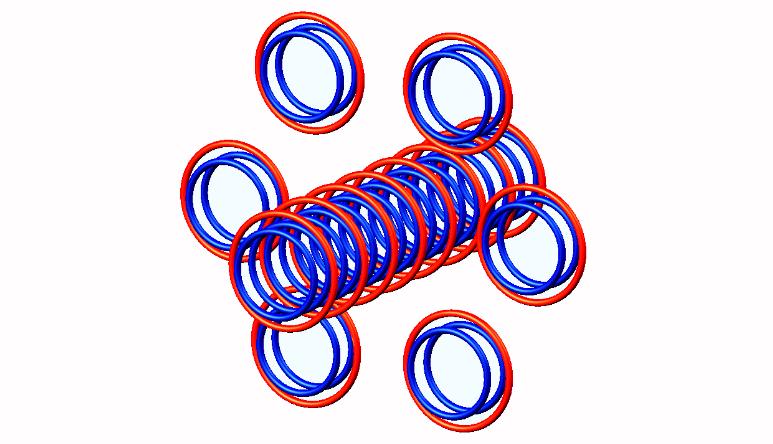
Nuclear structure of (stable). Move mouse on image for Silicon 29 (stable) nuclear structure
Blue toruses = 28 protons
Red toruses = 14 nuclear electrons
Atomic number of Si28 = protons (28) - nuclear electrons (14) = 14, stable with structure 0T+8D + 6D
Atomic number of Si29 = protons (29) - nuclear electrons (15) = 14, stable with structure 1T+7D + 6D
Atomic number of Si30 = protons (30) - nuclear electrons (16) = 14, stable with structure 2T+6D + 6D
The central rod of Si28 analogous with Oxygen 16 nucleus (stable). Si28 = O16 + 6 D
The central rod of Si29 analogous with Oxygen 17 nucleus (stable). Si29 = O17 + 6 D
The central rod of Si30 analogous with Oxygen 18 nucleus (stable). Si30 = O18 + 6 D
Click on image for Silicon 30 (stable) nuclear structure
Light photon model



Torus models of stable Silicon 28 nuclei. Blue torus = protons, red torus = nuclear bound electrons. Quarks and gluons do not exist.



