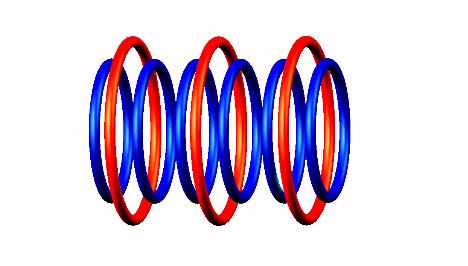
Nuclear structure of (stable) when mouse off the image. Move mouse on image for Lithium 7 (stable) nucleus
Blue toruses = 6 protons
Red toruses = 3 nuclear electrons
Atomic number of Li6 = protons (6) - nuclear electrons (3) = 3 with structure 3D.
Click on image for Berillium 7 nuclear structure (half life = 53.22 d), which transforms to Lithium 7 (stable) with K electron capture
Torus model of photon, proton, positron and electrons
Nuclear structure of elements



Nuclear atomic structure of isotope Lithium 6, stable and composition of Lithium 6 nuclei of stable element. Li6 -ot bombázzunk deutériummal. Be8-on keresztül He4 kell keletkezzen. Erős mágneses térben a hatás irányfüggő lehet?



