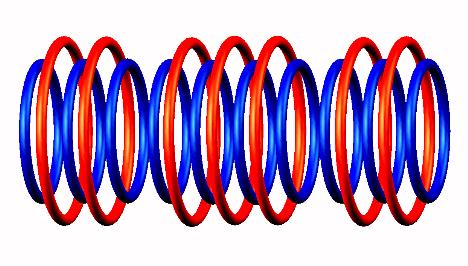
 destabilized form
destabilized form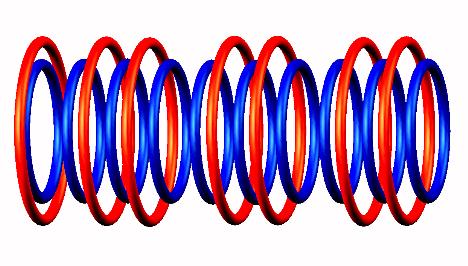 to Lithium 9
to Lithium 9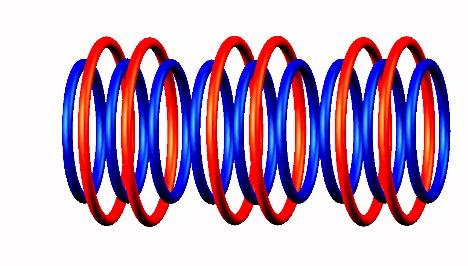 nucleus.
nucleus.
Blue toruses = 10 protons
Red toruses = 7 nuclear electrons
Atomic number of Li10 = protons (10) - nuclear electrons (7) = 3 with structure T H4 T.
Lithium 10 decays by neutron emission (from the left side) of
Lithium 10 destabilized form
destabilized form to Lithium 9
to Lithium 9 nucleus.
nucleus.
Lithium 9 decays by electron emission
(from the left side) of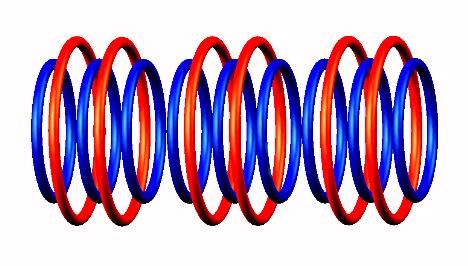 Lithium 9
Lithium 9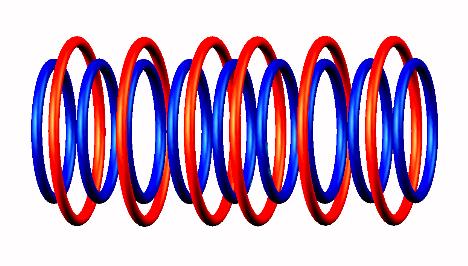 destabilized
destabilized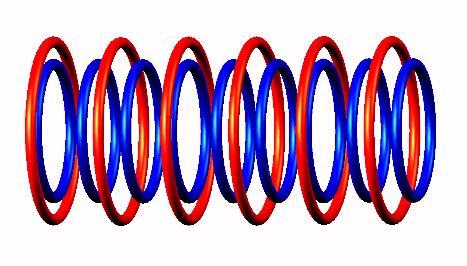 form
form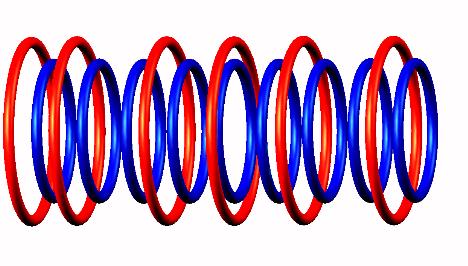 to Be 9
to Be 9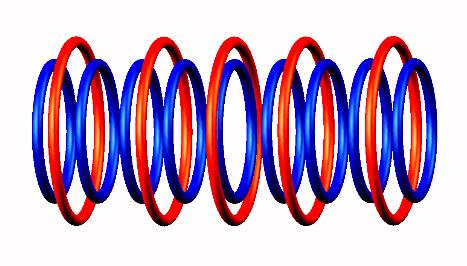 (stable) nucleus.
(stable) nucleus.
Click on image for Lithium 9 (unstable) nucleus.
Photon and matter
Structure of atomic nuclei



| Li4 | Li5 | Li6 | Li7 | Li8 | Li9 | Li10 | Li11 | Li12 |
| H | He | Li | Be | B | C | N | O | F | Ne | Na | Mg | Al | Si | P | S | Cl | Ar |
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | Pr | Nd | Sm | Gd | Tb | Dy | Ho | Er | Yb | Ta | W | Os | Ir | Au | Hg | Pb |
Neutron emission decay of Lithium 10 nuclear structure. Mechanism of Lithium 10 decomposition and production of Lithium 9 isotope. Nuclear torus model. The strong nuclear force is simply a close electromagnetic interaction between nuclear electrons and protons. There is no gluons in nuclei. Nor quarks. Neutron is simply a proton-electron torus pair. Neutrons are in stabilized condition inside nuclei by parallel proton toruses.