
 emission (from left and right side position) of Neon 16
emission (from left and right side position) of Neon 16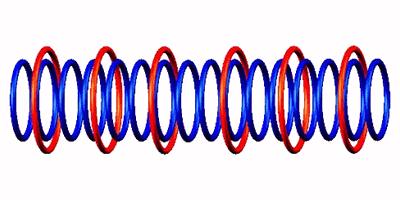 through destabilized form
through destabilized form 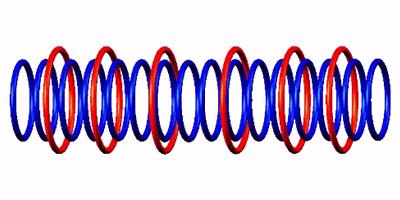 to Oxygen 14
to Oxygen 14 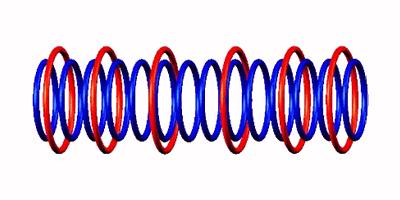 followed by
followed byOxygen 14 b+ transformation to Nitrogen 14
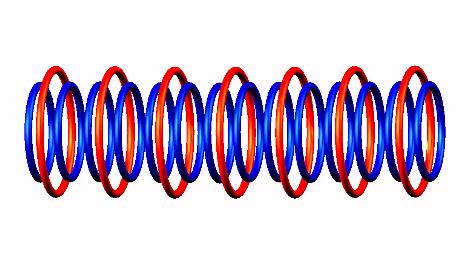 (stable), using photon.
(stable), using photon.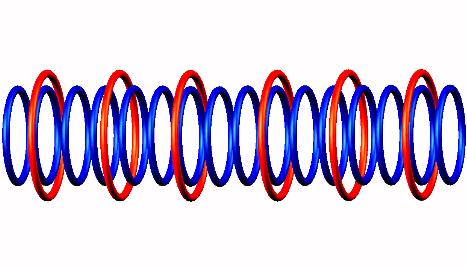
Blue toruses = 16 protons
Red toruses = 6 nuclear electrons
Atomic number of Ne16 = protons (16) - nuclear electrons (6) = 10 with structure 4p+6D = He3 D He3 He3 D He3.
Neon 16 decomposes by 2 proton
 emission (from left and right side position) of Neon 16
emission (from left and right side position) of Neon 16 through destabilized form
through destabilized form  to Oxygen 14
to Oxygen 14  followed by
followed by
Oxygen 14 b+ transformation to Nitrogen 14 (stable), using photon.
(stable), using photon.
Click on image for Oxygen 14 nuclear structure.
Photon and nuclear particle model
Light photon modell



| Ne16 | Ne17 | Ne18 | Ne19 | Ne20 | Ne21 | Ne22 |
| H | He | Li | Be | B | C | N | O | F | Ne | Na | Mg | Al | Si | P | S | Cl | Ar |
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf |
| Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi |
The nuclear composition of the Neon-16. Due to the very few bonding electrons, the Neon-16 nucleus decreases by 2 protons as each of the two outermost positions loses one proton.