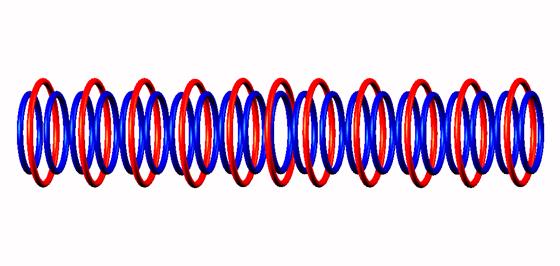
Nuclear structure of (stable).
Blue toruses = 21 protons
Red toruses = 11 nuclear electrons
Atomic number of Ne21 = protons (21) - nuclear electrons (11) = 10 with structure B10 n B10
Photon and particle model
Light photon modell
Neon 21 isotope nuclear composition, torus model of Neon 21.



The nuclear composition of the Neon 21 stable isotope. Blue toruses are protons, red toruses are the bonding electrons in the nucleus. There are no gluons, quarks or forth kind of strong nuclear force. Nor Higgs.



