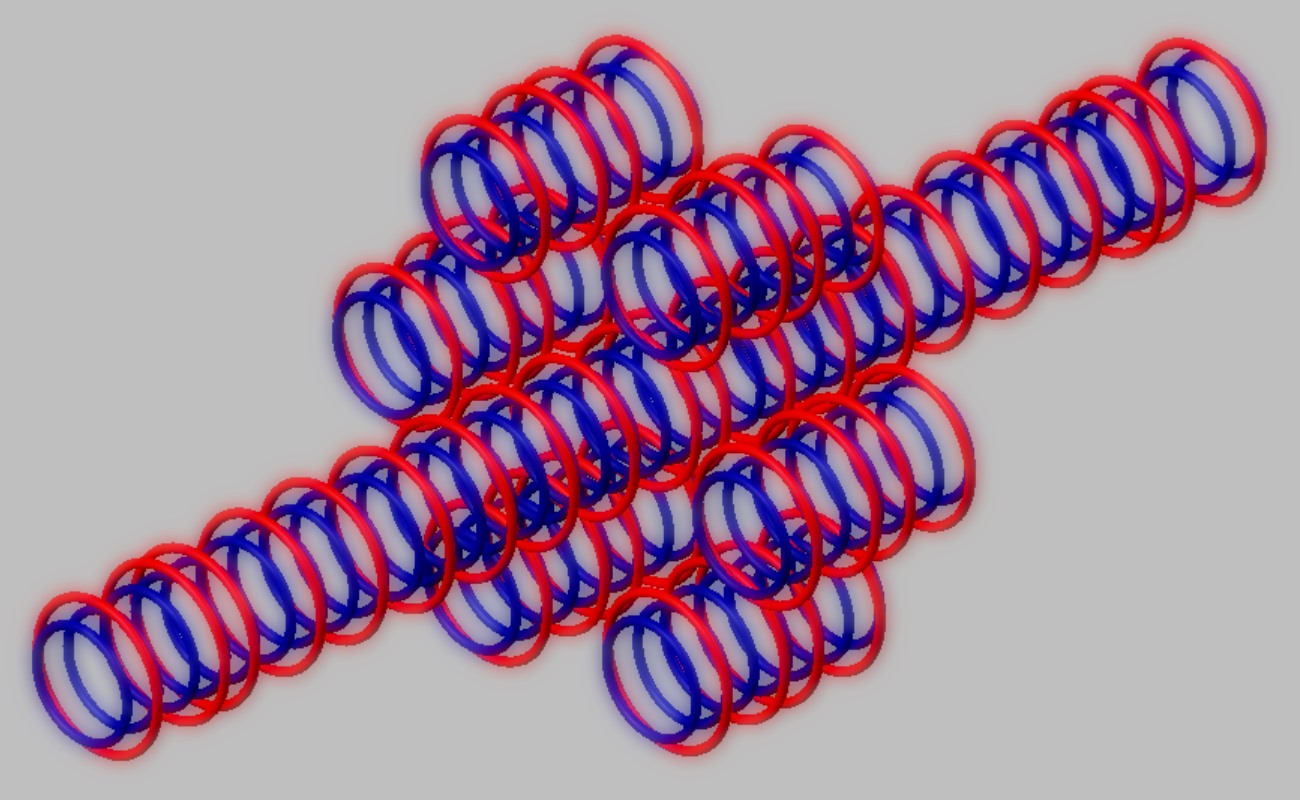
Nuclear structure of (stable).
Blue toruses = 79 protons
Red toruses = 44 nuclear electrons
Atomic number of Br79 = protons (79) - nuclear electrons (44) = 35 with structure 3T+14D + 6Li7
Atomic number of Br81 = protons (81) - nuclear electrons (46) = 35 with structure 5T+24D + 6T
The structure of Br79 central nanotube is 3T+14D corresponds to Cl37 stable nucleus.
The six member ring consists by 6 Lithium7 nuclei.
Click on image for bigger view of Br79 nucleus.
Torus model of photon and matter
Nuclear structure of the elements
Br 79



Nuclear structure of Bromine 79 (stable). Blue toruses = 79 protons. Red toruses = 44 nuclear electrons.



