


| Cr52 |
| H | He | Li | Be | B | C | N | O | F | Ne | Na | Mg | Al | Si | P | S | Cl | Ar |
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf |
| Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi |
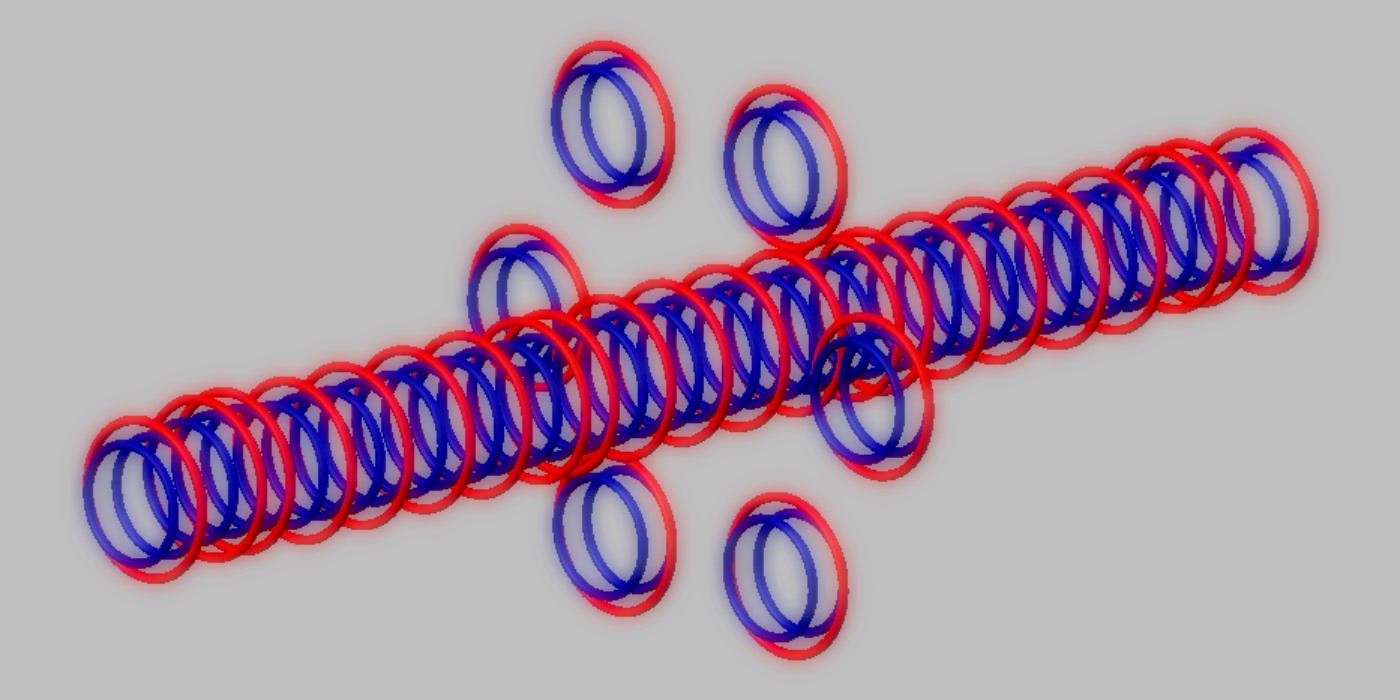
Blue toruses = 52 protons
Red toruses = 28 nuclear electrons
Atomic number of Cr52 = protons (52) - nuclear electrons (28) = 24 with structure 4T+14D + 6D.
Atomic number of Cr53 = protons (53) - nuclear electrons (29) = 24 with structure 5T+19D.
Central rod core of Cr52 stable nucleus image
= 4T+14D = 40 protons + 28 nuclear electrons (= Ar40 nucleus, stable). The ring = 6 D
Single rod nanotube of Cr53 = 5T+19D = 53 protons + 28 nuclear electrons, stable. No ring.
Click here for Cr52 stable nucleus image.
Light photon modell
A single Fe atom
Chromium Magnetism



| Cr52 |
| H | He | Li | Be | B | C | N | O | F | Ne | Na | Mg | Al | Si | P | S | Cl | Ar |
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf |
| Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi |
Nuclear structure of stable Chromium 52 isotope.