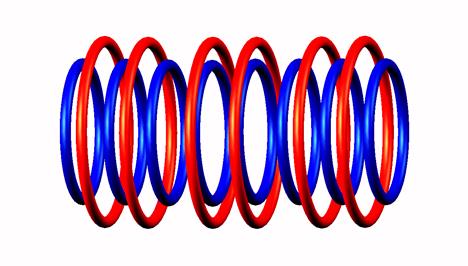
 destabilized,
destabilized, 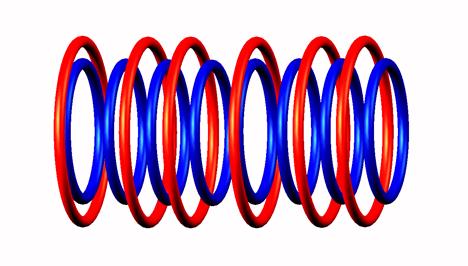 decay form
decay form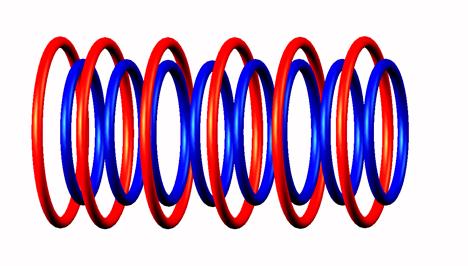 to Lithium 8
to Lithium 8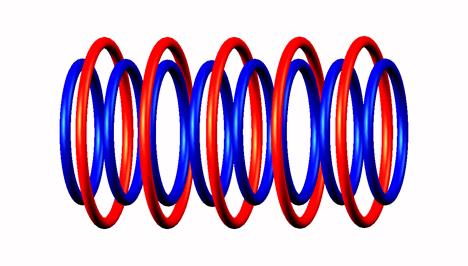 .
.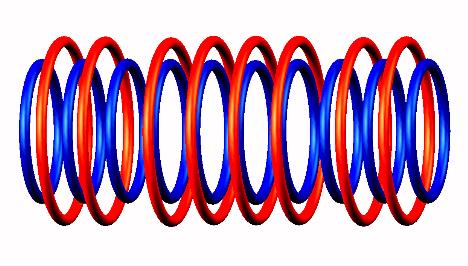
Blue toruses = 10 protons
Red toruses = 8 nuclear electrons
Atomic number of He10 = protons (10) - nuclear electrons (8) = 2 with structure T n n n n T.
Helium 10 decays by 2 neutron emission of Helium 10 neutron excess to Helium 8
Helium 8 decays by electron emission (from the left position) of
Helium 8  destabilized,
destabilized,  decay form
decay form to Lithium 8
to Lithium 8 .
.
Lithium 8 decays by electron emission of
Lithium 8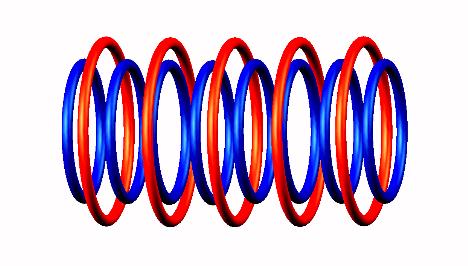 destabilized form
destabilized form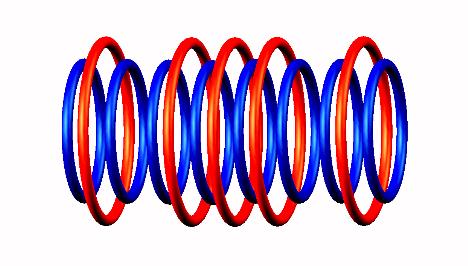 through Be8
through Be8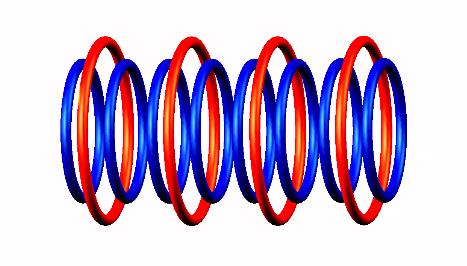 producing two He4
producing two He4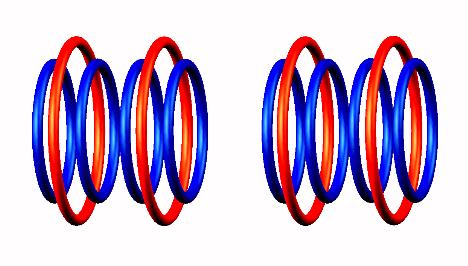 nucleus (stable).
nucleus (stable).
(The neutrons and the subsequent electron emission really happens from the end of the nuclear nanotube, NOT from the central position. Im just depict central neutrons for the sake of picturesque symmetry.)
Click on image for Helium 8 nucleus
Yin-yang model of photon and matter
Structures of atomic nuclei of elements



| He3 | He4 | He5 | He6 | He7 | He8 | He9 | He10 |
| H | He | Li | Be | B | C | N | O | F | Ne | Na | Mg | Al | Si | P | S | Cl | Ar |
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | Pr | Nd | Sm | Gd | Tb | Dy | Ho | Er | Yb | Ta | W | Os | Ir | Au | Hg | Pb |
Helium 10 double neutron emission and mechanism of He10 isotope decay. The subsequent electron emission of destabilized He8 produce Li8 which emits an electron and the Be8 split into two He4 alpha particles.