 to Nitrogen 17
to Nitrogen 17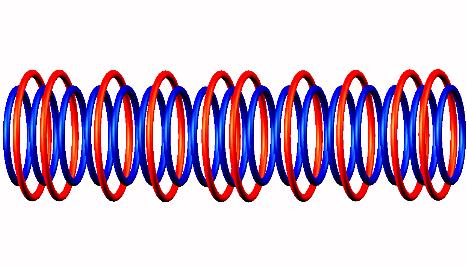 (half life = 4.173 s).
(half life = 4.173 s).
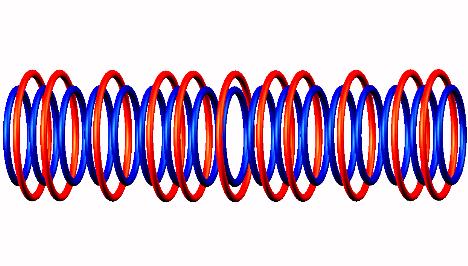
Blue toruses = 17 protons
Red toruses = 11 nuclear electrons
Atomic number of C17 = protons (17) - nuclear electrons (11) = 6 with structure TDTnTDT or 5T+1D.
Carbon 17 decays by electron emission (from a side position, not indicated) of Carbon 17 to Nitrogen 17
to Nitrogen 17 (half life = 4.173 s).
(half life = 4.173 s).
Nitrogen 17 decays by electron emission (from a side position, not indicated) of
Nitrogen 17 to transitional Oxygen 17
to transitional Oxygen 17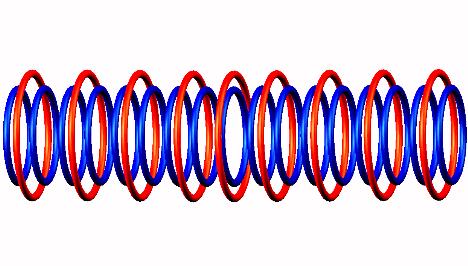 and
and
subsequently a neutron emission of transitional (activated) Oxygen 17 decay form
decay form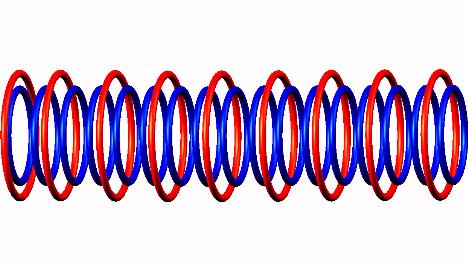 producing Oxygen 16
producing Oxygen 16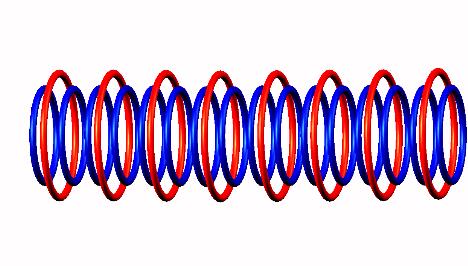 stable nucleus.
stable nucleus.
Click on image for Oxygen 16 nucleus.
Torus model of photons and matter
Structure of atomic nuclei



| C8 | C9 | C10 | C11 | C12 | C13 | C14 | C15 | C16 | C17 | C18 | C19 |
| H | He | Li | Be | B | C | N | O | F | Ne | Na | Mg | Al | Si | P | S | Cl | Ar |
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf |
| Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi |