the electron captured to central part of Carbon 9
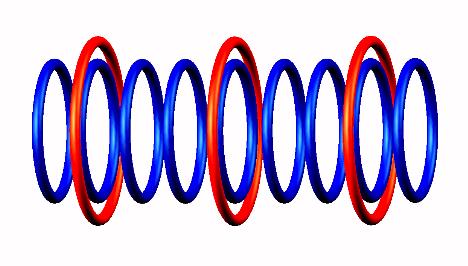 forming Boron 9
forming Boron 9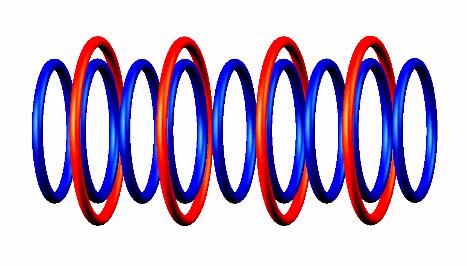 nucleus.
nucleus.
Blue toruses = 9 protons
Red toruses = 3 nuclear electrons
Atomic number of C9 = protons (9) - nuclear electrons (3) = 6 with structure He3 He3 He3.
Carbon 9 emits positron from a source of photon decomposition, while other component of the photon,
the electron captured to central part of Carbon 9 forming Boron 9
forming Boron 9 nucleus.
nucleus.
Boron 9 destabilized form
destabilized form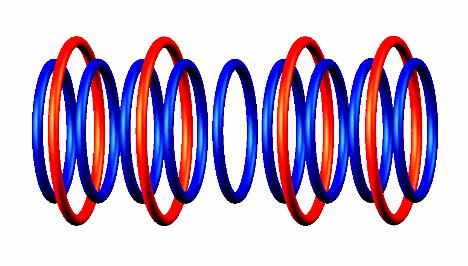 decays
decays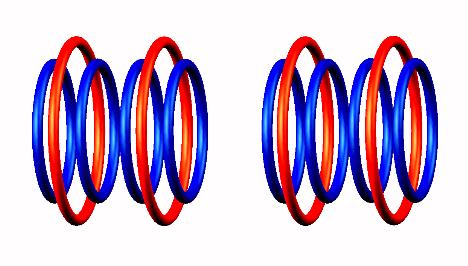 by
proton emission (uniquely from central position), forming two He4 nucleus particles.
by
proton emission (uniquely from central position), forming two He4 nucleus particles.
Carbon 9 may formed by decay of Nitrogen 10 nucleus
Photon model of light
Structure of nuclei of elements
Nuclear symmetry



| C8 | C9 | C10 | C11 | C12 | C13 | C14 | C15 | C16 | C17 | C18 | C19 |
| H | He | Li | Be | B | C | N | O | F | Ne | Na | Mg | Al | Si | P | S | Cl | Ar |
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf |
| Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi |