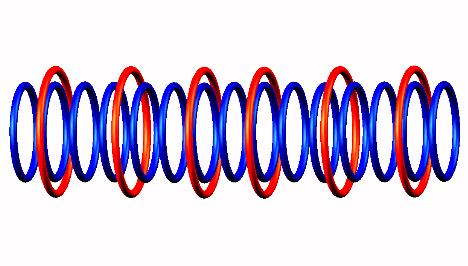
 decay form
decay form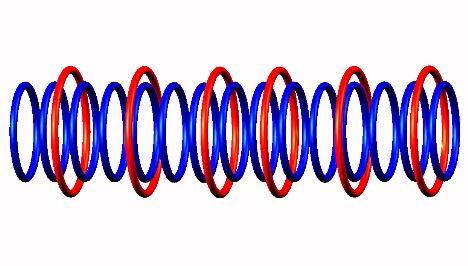 to Oxygen 14
to Oxygen 14 .
.
Blue toruses = 15 protons
Red toruses = 6 nuclear electrons
Atomic number of F (9) = protons (15) - nuclear electrons (6) = 9 with structure 3p+6D = He3 D Li5 D He3.
Fluorine 15 nucleus transform by proton emission (left side) of
Fluorine 15 decay form
decay form to Oxygen 14
to Oxygen 14 .
.
Oxygen 14 transformed by positron emission from a source of a photon decomposition, while
electron component of the photon captured to the center of
Oxygen 14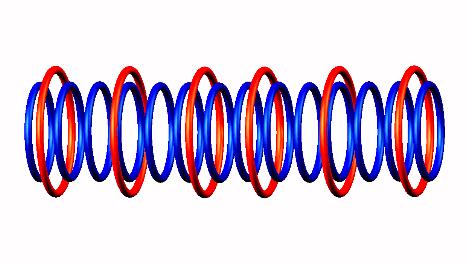 decay form,
decay form, 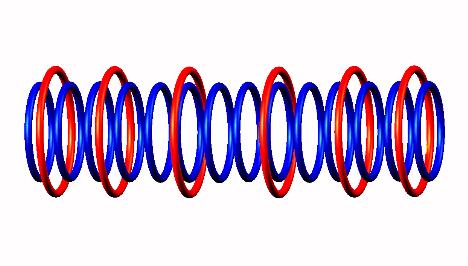 generating Nitrogen 14
generating Nitrogen 14 (stable).
(stable).
Click on image for Oxygen 14 nuclear structure.



| F14 | F15 | F16 | F17 | F18 | F19 | F19i |
| H | He | Li | Be | B | C | N | O | F | Ne | Na | Mg | Al | Si | P | S | Cl | Ar |
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf |
| Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi |
Fluor-15 transforms with proton emission from one end of the nuclear nanotube.