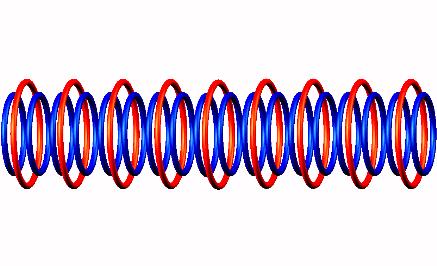
Nuclear structure of (half life = 109.77 min).
Blue toruses = 18 protons
Red toruses = 9 nuclear electrons
Atomic number of F18 = protons (17) - nuclear electrons (8) = 9 with structure 9D.
Fluorine 18 transform by positron emission from a source of a photon decomposition,
while electron component of the photon captured by
Fluorine 18 and Oxygen 18 produced (stable).
Click on image for Fluorine 18 nuclear structure.
Light photon modell



The nuclear composition of the Fluor-18 isotope, nuclear structure of the F18 atom, nucleus of the Fluor-18 element. Positron emission decay of Fluorine-18 with gamma photon capture.
The structure of F18 nucleus appears balanced and symmetrical, yet it is still electron-deficient. Presumably, extremely high pressure stabilizes these longer structures in a neutron star.



