 decay form,
decay form,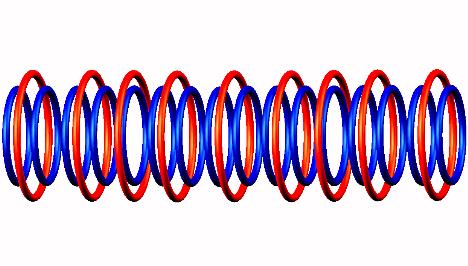 forming Oxygen 16
forming Oxygen 16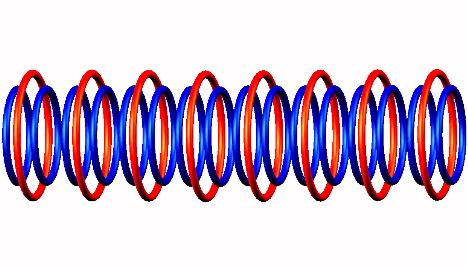 nucleus (stable).
nucleus (stable).
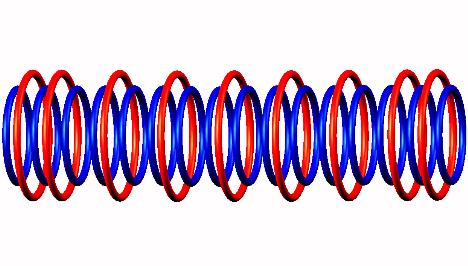
Blue toruses = 16 protons
Red toruses = 9 nuclear electrons
Atomic number of N16 = protons (16) - nuclear electrons (9) = 7 with structure 2T+5D.
Nitrogen 16 decays by electron emission of
Nitrogen 16 decay form,
decay form, forming Oxygen 16
forming Oxygen 16 nucleus (stable).
nucleus (stable).
In reality the emission of an electron always happen from one of the end position of proton nanotube (not indicated), not from central part, never.
Click mouse on image for Oxygen 16 nucleus.
Yin-yang photon model of light
Nuclear symmetry,
Nuclear structure of elements



| N10 | N11 | N12 | N13 | N14 | N15 | N16 | N17 | N18 | N19 |
| H | He | Li | Be | B | C | N | O | F | Ne | Na | Mg | Al | Si | P | S | Cl | Ar |
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf |
| Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi |
Nitrogen 16 nuclear structure, half life = 7.13 sec.