 decay form
decay form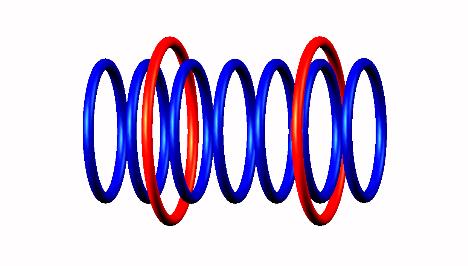 producing Be6 nucleus
producing Be6 nucleus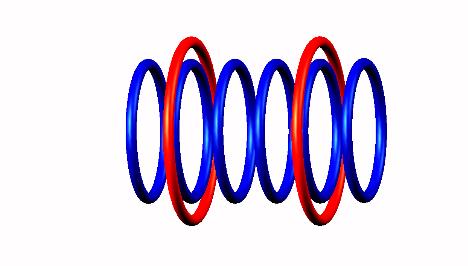 (half life = 5 zs)
(half life = 5 zs)
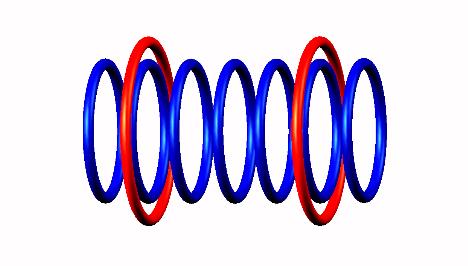
Blue toruses = 7 protons
Red toruses = 2 nuclear electrons
Atomic number of B7 = protons (7) - nuclear electrons (2) = 5 with structure He3 p He3.
Boron 7 nucleus decays as highly instable with proton emission (from left side position) of Boron 7 nucleus decay form
decay form producing Be6 nucleus
producing Be6 nucleus (half life = 5 zs)
(half life = 5 zs)
Beryllium 6 decays by 2 proton emission (left and right side protons) of
Beryllium 6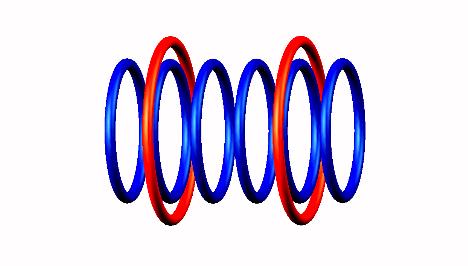 destabilized form
destabilized form and He4 nucleus
and He4 nucleus produced.
produced.
Click on image for Be6 nucleus
Photon model of light
Structures of atomic nuclei



| B7 | B8 | B9 | B10 | B11 | B12 | B13 | B14 | B15 | B16 | B17 | B18 |
| H | He | Li | Be | B | C | N | O | F | Ne | Na | Mg | Al | Si | P | S | Cl | Ar |
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr |
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe |
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf |
| Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi |